The COVID-19 pandemic is affecting health care practitioners of all levels with an expectation of a rapid increase in case numbers outstripping departmental resources. Colleagues around the world are reporting Lung Ultrasound as being highly useful for triage, diagnosis and assessing response to management in affected patients.
This post is meant as a quick guide for those on the front line based on early reports from the international community. Rapid up-skilling in POCUS now will dramatically improve diagnostic potential – vital in the coming weeks.
Lung ultrasound has a better diagnostic accuracy than chest X-rays and is equivocal to chest CT in many cases – provided the pathology extends to the pleura. Performing this at the bedside reduces risk of further spread by transporting patients to radiology and rapidly increases speed to diagnosis.
Infection control measures are key:
This should go without saying – moving a single machine between patients is an obvious source of cross contamination and users must be diligent to clean before and after use (never trust it is clean!).
Consider using a probe cover if available and if not a dedicated ‘COVID machine’. We have two machines on our COVID ITU which are not used for other patients but I accept other departments are not as fortunate.
Remember the basics:
As ever with POCUS – remember to orientated yourself, optimise images, record for quality assurance and don’t overcall findings. Rule in not rule out.
If new to lung US there are plenty of resources out there and consider finding a mentor ASAP to guide you through the process.
Some resources:
- Great article by Dr Ashley Miller in BJA – Practical approach to lung ultrasound.
- An update on the original article on lung US by Lichtenstein.
- E-LFH – ICE-BLU e-learning
- E-LFH – FAMUS e-learning
- Plenty of others – twitter/youtube etc.
Use linear probe to assess pleura (relevant in COVID).
Curvilinear or phased for lung windows.
Link to FUSIC mentors and FAMUS mentors.
Quick reminder of the classic signs:



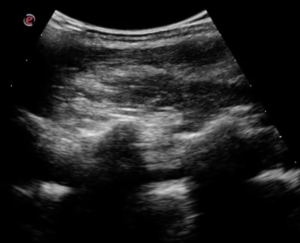
Note the difference between the normal thin, regular pleural line and the thickened, irregular line on the right.

COVID-19
Key findings seen in COVID-19 patients are as follows (link to original article):
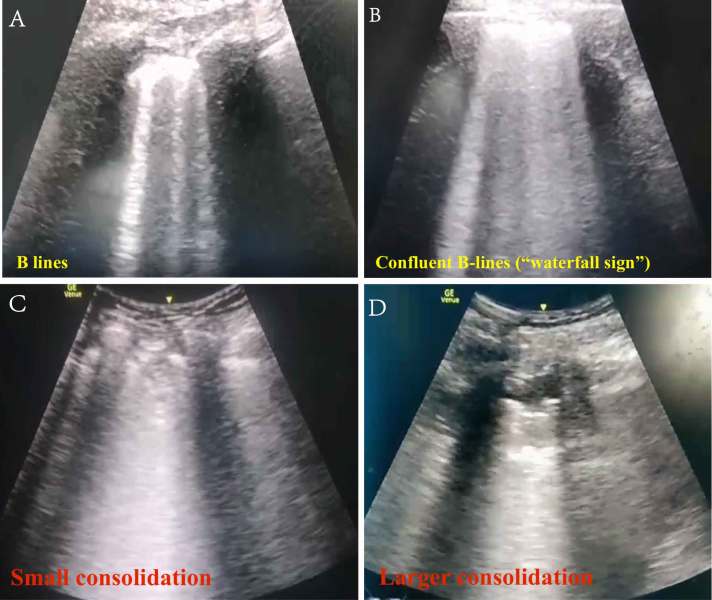
- Interstitial syndrome pattern – extensive B lines which may coalesce. Key is they may be distributed in any pattern with clear skips between the lesions (patches of B-lines with thickened pleura interspersed with normal lung).
- Thickening of the pleural line with irregularity.
- Subpleural consolidation may be seen alongside the pleural thickening.
- Other patterns of consolidation – small, non-translobar, translobar, air-bronchograms.
- A-lines during recovery.
- Pleural effusions are rare.
It is worth noting that interstitial syndrome pattern is not unique to COVID-19. By far the most common cause in routine practice is pulmonary oedema (cardiogenic/renal) and follows a continuous course ascending up the chest with disease severity.
COVID-19 findings mimic those seen in other causes of interstitial pneumonitis/acute respiratory distress syndrome (ARDS) but may present significantly earlier in the disease process. Patients may be clinically well with LUS findings. Dr Yale Tung Chen, an ED physician in Madrid, has a brilliant twitter thread documenting his own lung US findings after being diagnosed with COVID-19.
Day 6 after #COVID diagnosis. Less cough, bit tired, still no dyspnea. No fever. Oxygen saturation 98%. #POCUS update: thick pleural line, b-lines and subpleural consolidations tend to resolve. Significant lung improve from yesterday. #mycoviddiary @TomasVillen @acam_acam pic.twitter.com/StQnbPPFUE
— Yale Tung Chen (@yaletung) March 14, 2020
In patients presenting with symptoms suggestive of COVID-19 and an interstitial syndrome pattern on lung US (B-lines etc) a positive diagnosis is highly likely.
Regular US can be used to track disease progression. Lessons from our Italian colleagues (summarised by Massimo Gianni) have divided findings into two distinct patterns:
- Pattern 1 – diffuse B-lines – in ventilated patients this may respond well to increased PEEP.
- Pattern 2 – consolidation, parapneumonic effusions and atelectasis. If significant posterior atelectasis in ventilated patient they may respond well to pronation.
- Reassessment can assess response to increased PEEP or recruitment manoeuvres, new consolidation in superimposed bacterial infection.
- Identify complications of high airway pressures/recruitment manoeuvres e.g. pneumothorax/surgical emphysema.
The Butterfly Inc website has a good page – Fighting COVID-19 Together – which has a few examples of the findings with an explanation.
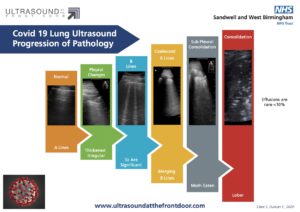
Peng et al suggested the differing features may correlate with disease severity on CT:
- Least severe – scattered B-lines correlates to mild ground-glass opacity on CT.
- Coalescent B lines – more confluent ground-glass opacity.
- Peripheral consolidation on CT and US with more severe disease.
- Most severe disease – more consolidated lung.
Example lung images & progression from Springer Link:
Finally:
Findings for COVID-19 are fairly straight forward and the skills can be acquired rapidly and easily. Identify a FUSIC/FAMUS mentor in your hospital, read some background resources and do the e-learning (it doesn’t take very long).
It is going to be a difficult time for everyone in hospitals and resources are going to be dramatically stretched. Learning these skills will save time and limit unnecessary additional investigations – not to mention empowering you as frontline clinicians.
Even prior to completing accreditation this may be used sensibly provided someone can review images and it isn’t used to ‘rule out’ pathology. Most hospitals will now have mentors available and online resources are available for remote supervision – e.g. www.sonoclipshare.com.
Don’t forget assessment of other systems too – ~7-10% incidence of myocarditis in severe cases which is an easy pickup and may significantly complicate management.
References:
Twitter – usual suspects @paradicmshift @icmteaching @wilkinsonjonny @youricm
Dr Yale Tung Chen’s great twitter threat.
Miller, A. Practical Guide to Lung Ultrasound. British Journal of Anaesthesia Education (2016)


What about the cooling fan system on most cart and laptop systems? Any concern about viral spread and inability to disinfect these components? I doubt their filters are designed to halt virus sized particles. Should only fanless handheld systems be used?
Interesting thought. Ours doesn’t have large fan vents but there are many moving parts in all of the machines. Probe covers and keeping a distance from the patient is probably the best we can do as things get worse. Have seen people put their phone & portable US all within a probe cover to contain the entire device.